Chemical Quality Management Software
QMS Software for Chemical Industries
In Chemical manufacturing, it is essential that your Quality Management System enables effective and easy tracking. Our Quality Management software for Chemical Industries enables you to capture data from a wide range of operational processes and share that content so that your biggest concerns are addressed.
- Deliver actionable information.
- Reduce manual tasks.
- Lower materials costs.
- Support compliance needs.
Turning Data into Information for Chemical Manufacturers
For manufacturers in chemical and aligned industries, the integration of people, equipment and products generates a flood of data. To be useful and relevant, this data needs to be managed so that actionable events are identified, targeted to the right destination, and monitored to ensure prompt action. Moving seamlessly from task to task with the right information at your fingertips delivers greater efficiency and control of outcomes, from materials use to timely documentation.
Inputs
- Collect data from anywhere in your process.
- Use Set up records to support repeatable tasks
- Automate receiving by importing Vendor COA data.
- Import Equipment generated testing results.
Outputs
- Establish automatic notifications for out of range events.
- Provide analytical data without additional transcription or manipulation.
- Share process performance metrics in real time.
- Automate delivery of reports to reduce throughput times.
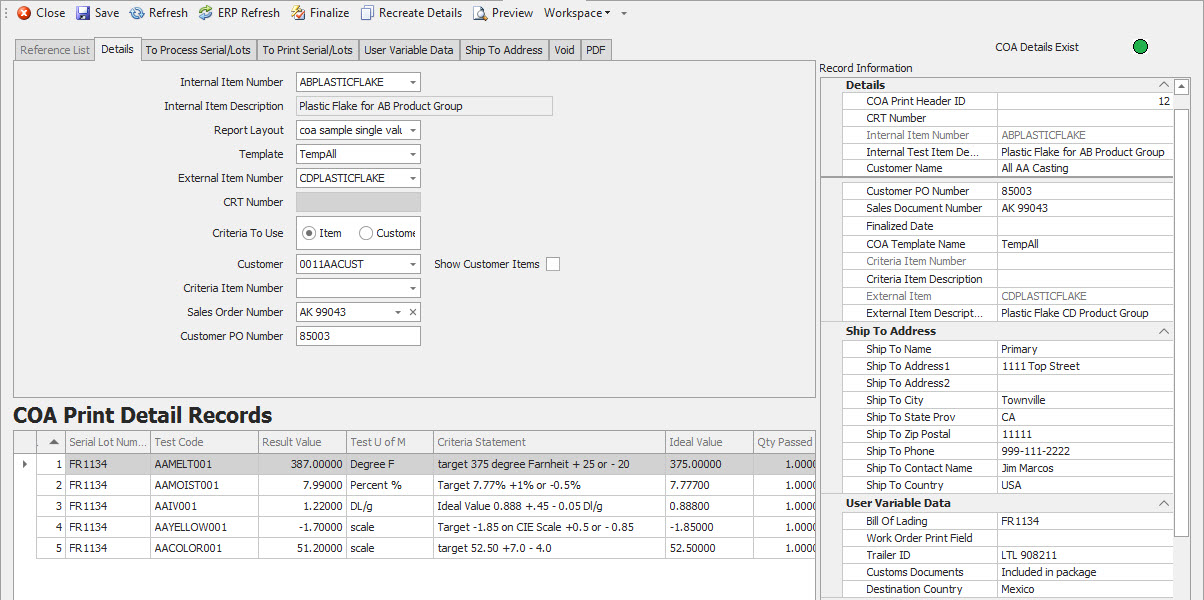
Efficient Generation of Certificates of Analysis in Chemical Manufacturing
One of the most time-sensitive tasks for chemical manufacturers is ensuring that valid and accurate Certificates of Analysis are available on-time to support every shipment. Customer specifications for data on these documents can vary significantly and failure to accompany shipments with the correct document can lead to rejected shipments and refusal to pay.
We make the task of generating Certificates of Analysis easy with 4 key features.
- Item and Lot parameters search stored data to easily identify required data set.
- Data is filtered precisely to meet customer specifications with preconfigured data templates.
- Quick reference records store repeatable default selections for COA inputs to avoid rekeying errors.
- Unlimited Report layout records enable accurate data presentation for paper documents.
An Affordable Chemical Quality Management Solution
The Benefit of a Single License Supporting Unlimited Concurrent User Access
Let's face it... no matter how beneficial digital transformation is for you company, if it is too costly it won't happen. Most cost concerns come down to the number of licenses to buy that will still let you get the job done; not done efficiently, just done.
By providing unlimited user access we make sure you can:
- Define YOUR most efficient processes.
- Enable collaboration inside and outside of your organization.
- Keep cost of growth predictable, adding users doesn't mean buying more seats.
Get Started Now
Contact Information
SALES
sales@qualityessentialssuite.com
+1 (866) 949-9504, ext 811
SUPPORT
support@qualityessentialssuite.com
+1 (866) 949-9504, ext 2

